Auteur: Paweł Radzikowski radzikpawe@gmail.com

Dag 1 van de controle (29.04.2024): Bij aankomst in het Duitse Living Lab ontmoette de heer Paweł Radzikowski van Ogolnopolskie Stowarzyszenie Agrolesnictwa (OSA, Poolse Agroforestry Vereniging) mevrouw Janine Raabe, de boerin van Hof Lebensberg. De precieze locaties voor de bemonsteringslocaties werden bepaald en er werden controlelocaties aangewezen. De onderzoeken werden uitgevoerd in twee agrobosbouwsystemen: gemengd-fruitsystemen en gemengd-notensystemen. De controle voor het gemengde notensysteem was een aangrenzend veld met een voedergewas, terwijl het gemengde vruchtensysteem een aangrenzend, onbeplant veld was. Tijdens de besprekingen ter plaatse werd besloten om het bemonsteringsprotocol dat oorspronkelijk in taak 3.2 was ontwikkeld, aan te passen aan de omstandigheden van de locatie. In plaats van een gradiënt met vier afstanden tot de bomen, werd gekozen voor twee afstanden: één direct naast de bomen en één halverwege tussen de bomen. Deze aanpassing was nodig omdat de bomen pas een jaar of twee voor het begin van de monitoring waren geplant, waardoor hun impact vooral op korte afstand merkbaar was. Indirecte afstanden werden in deze context niet als significant beschouwd.
In beide systemen en hun respectieve controles werden testpercelen van 30 x 10 meter ingericht. De lengte van 30 meter werd gekozen omdat voor bepaalde biodiversiteitsmonsters de locaties minstens 10 meter uit elkaar moesten liggen. Ervan uitgaande dat vier herhalingen gewenst waren voor elke variant, zouden deze langs één transect worden geplaatst op afstanden van 0m, 10m, 20m en 30m. De breedte van 10 meter werd gekozen vanwege de afstand van 20 meter tussen de bomenrijen. De bemonstering werd zo gepland dat het transect langs de bomen liep en het midden van het akkerbouwveld bereikte (zie Fig. 1 tot 3). Dezelfde opstelling werd toegepast op het controlegebied, hoewel het veld overal uniform was.
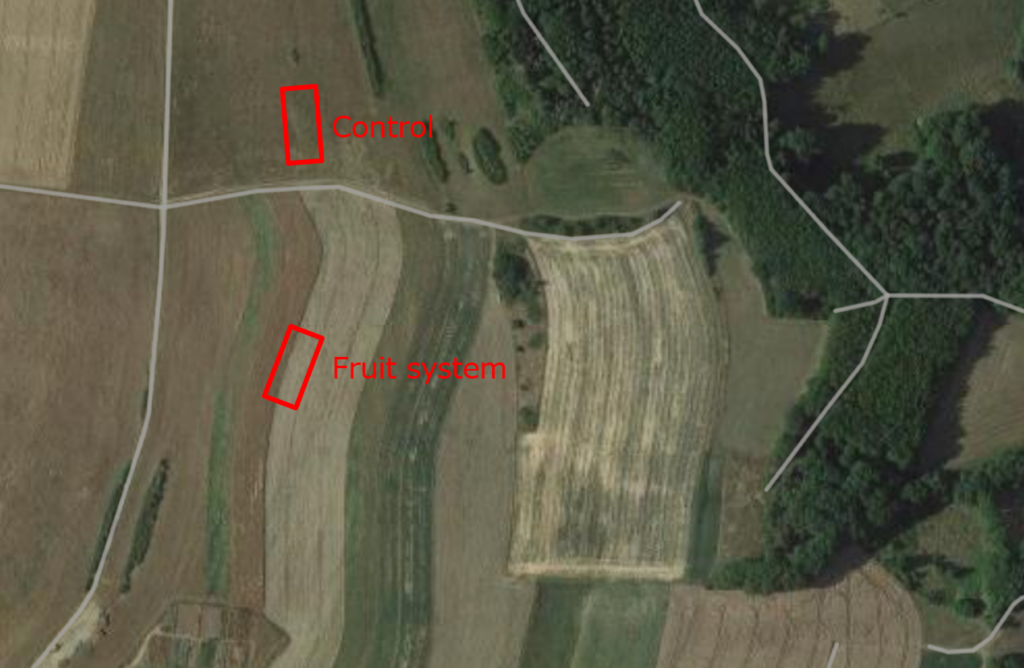
Fig. 2 Testpercelen in het gemengde vruchtensysteem en de controle

Fig. 3 Precieze locatie van de bemonsteringslocaties in het gemengde moerassysteem

Fig. 4 De lengte en breedte van het testveld meten
De monitoring begon met de minst invasieve methoden, namelijk het verzamelen van insecten. Dit onderzoek werd uitgevoerd over drie transecten van 10 meter in elk studieperceel (Fig. 5). Er werd een speciaal entomologisch net gebruikt met een steel van 1 meter en een hartvormige opening van 30 x 40 cm. Dit ontwerp vergemakkelijkte het verzamelen van insecten van zowel hoge vegetatie als oppervlakken dichtbij de grond. De opvangzak van het net is gemaakt van fijn gaas versterkt met een stevige materiaalhoes (Fig. 6). De insecten die tijdens het onderzoek verzameld werden, werden overgebracht in bakjes van 100 ml, geëuthanaseerd en geconserveerd met ethanol 75% (Fig. 7). In totaal werden 36 monsters verzameld en de identificatie van deze insecten zal later worden uitgevoerd in taak 3.2.

Fig. 5 Locatie van insectenverzamelingstransecten

Fig. 6 Entomologisch net in gebruik

Fig. 7 Monsters verzameld met een entomologisch net
Op de eerste dag van de monitoring was een andere taak het uitzetten van vallen voor het verzamelen van insecten gedurende de volgende vijf dagen. Er werden twee soorten vallen gebruikt: Barber-aardevallen en gele bestuiversvallen. Deze vallen werden om de 10 meter geplaatst langs de aangewezen transecten, met drie vallen per variant (Fig. 8). De Barber-aardevallen, met een diameter van 10 cm, waren ontworpen om effectief bodeminsecten te vangen. De gele bestuiversvallen, met een diameter van 30 cm, werden direct op de grond geplaatst vanwege de lage vegetatiehoogte. Beide types vallen werden voor tweederde gevuld met een oplossing van zuiver water en geurloos detergent (Fig. 9). Deze oplossing zorgde ervoor dat de ongewervelden snel verdronken en dat ze niet overliepen tijdens hevige regenval, zonder dat de vallen afgedekt hoefden te worden gezien de korte verzamelperiode. Het waterniveau werd dagelijks aangevuld om volledige verdamping te voorkomen.
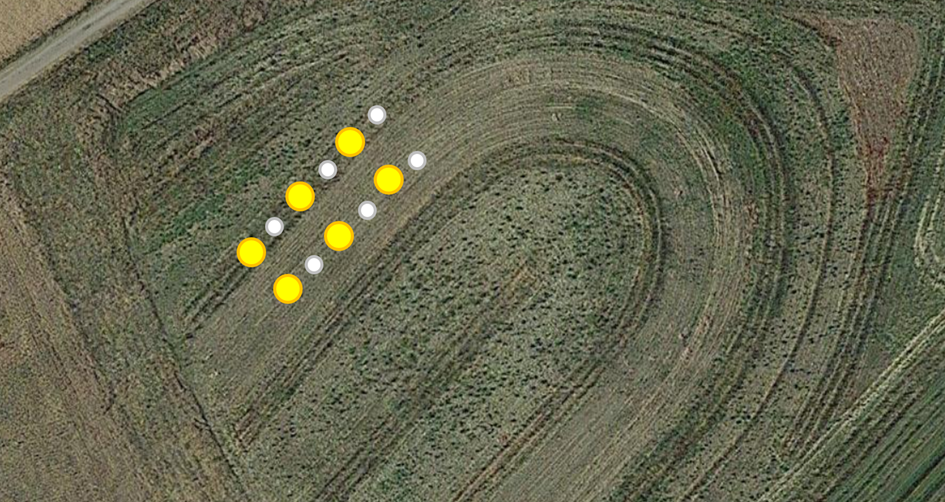
Fig. 8 Locatie van Barber-vallen en gele schalen in het gemengde notensysteem

Fig. 9 Het proces van het zetten van gele schalen en Barbervallen
Dag 2 van monitoring (23.04.2024): De beoordeling richtte zich op de biodiversiteit van de flora en de primaire productie. Binnen elk van de acht transecten werden vier punten geselecteerd voor fytosociologische foto's met behulp van een botanisch frame van 1×1 meter (Fig. 10). Alle plantensoorten werden geïdentificeerd en hun abundantie werd geschat volgens de aanbevolen veldprotocolrichtlijnen. De gegevens werden genoteerd op veldformulieren (Fig. 11).

Fig. 10 Verdeling van biodiversiteit en biomassabemonstering in het gemengde notensysteem

Fig. 11 Botanisch kader en veldformulier gebruikt bij de florabeoordeling
Biomassa binnen dezelfde meterkaders werd gesneden, geoogst en gewogen (Fig. 12). Hierdoor kon de opbrengst van voedergewassen voor grazende kippen worden geschat of de natuurlijke productiviteit van het ecosysteem (primaire productie) worden beoordeeld als de biomassa niet was geoogst. Ten tijde van de monitoring werd er geen opbrengst verkregen van de aanwezige bomen, noch werden de graanvelden ingezaaid. Daarnaast werd er fotografische documentatie van individuele plantensoorten in elk systeem en hun controle uitgevoerd (Fig. 13). Deze foto's werden geüpload naar iNaturalist voor verificatie van de soortidentificatie, waarbij gebruik werd gemaakt van algoritmen en overleg met specialisten. Waarnemingen werden geotagged om biodiversiteitsvergelijkingen wereldwijd te vergemakkelijken (Fig. 14).

Fig. 12 Geoogste voederbiomassa per 1 m2

Fig. 13 Individuele fotografische documentatie van planten
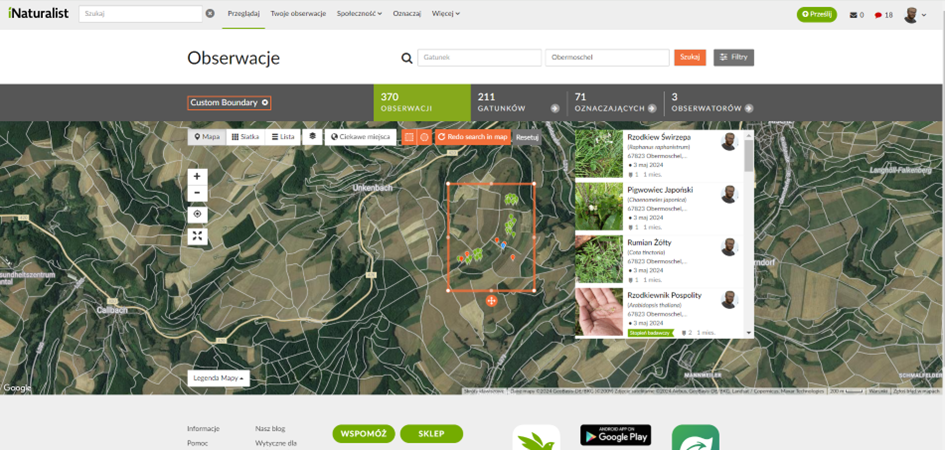
Fig. 14 Waarnemingen ingevoerd op iNaturalist
Dag 3 van monitoring (01.05.2024) - De activiteiten waren gericht op het onderzoeken van de bodemdiepte en het verzamelen van regenwormen. Bodemblokken van 25 x 25 x 25 cm werden uitgegraven op plaatsen waar de vorige dag biomassa was verzameld (Fig. 15). De grond werd voorzichtig op een plaat gescheiden en verder uitgegraven om bij de ondergrond te komen. Voor diepere bodemlagen werd indien nodig een Egner monsternemer gebruikt (Fig. 16).

Fig. 15 Verzamellocaties regenwormen en bodemdieptemetingen

Fig. 16 Meting van de bodemdiepte
De grond die op het zeil werd uitgespreid, werd handmatig gezeefd om regenwormen eruit te halen (Fig. 17). De gevonden exemplaren werden bewaard in met 75% alcohol gevulde bakjes om ze vervolgens te wegen en de soort te identificeren (Fig. 18).

Fig. 17 Grondblok doorsnijden en grond zeven voor regenwormen

Fig. 18 Extractie van regenwormen
Dag 4 van controle (2 mei 2024) - De bulkdichtheid en permeabiliteit van de bodem werden gemeten. De monsters werden in drievoud genomen op elke aangewezen locatie (Fig. 19). Voorafgaand aan de bemonstering werden kleine stukken graszode vrijgemaakt met een spade. Voor deze metingen werden twee soorten cilinders gebruikt. Kleine cilinders met een diameter van 5 cm werden in de grond gedreven om monsters te nemen voor bulkdichtheid (Fig. 20). De monsters werden verzegeld met deksels aan beide uiteinden en zullen later naar het bodemlaboratorium worden gestuurd voor analyse.
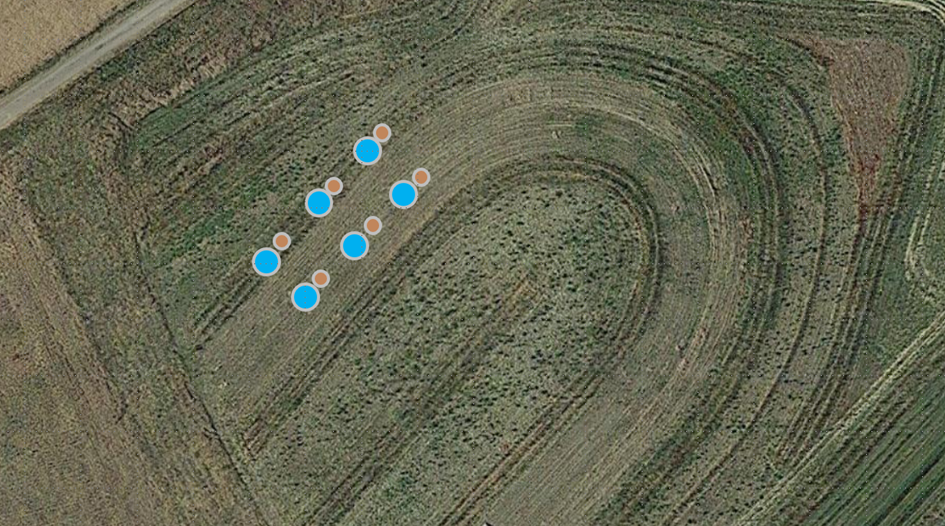
Fig. 19 Schematische weergave van het verzamelen van grondcilinders en doorlatendheidstesten

Fig. 20 Cilinderafname voor bulkdichtheid van grond
De waterdoorlatendheid van de bodem werd beoordeeld met behulp van cilinders met een diameter van 10 cm die tot de volledige diepte in de bodem werden gedreven, waarbij 2,5 cm werd vrijgelaten met een houten stop (Fig. 21). Vervolgens werd 100 ml water in de cilinder gegoten en werd de totale infiltratietijd gemeten met een stopwatch. Deze metingen werden genoteerd op de veldgegevensformulieren.

Fig. 21 Bodemdoorlatendheidsmeting
Dag 5 van toezicht (3 mei 2024) - De nadruk lag op het voltooien van het verzamelen van insectenvallen en het nemen van bodemmonsters. Insectenmonsters uit de gele schalen en bodemvallen werden gezeefd en overgebracht naar afgesloten containers met 75% ethanol (Fig. 22). De insectensoorten zullen in een later stadium worden geïdentificeerd.

Fig. 22 Verzamelen van insectenvallen
Bodembemonstering volgde de richtlijnen van veldprotocol Taak 3.2, met vier monsters verzameld in plastic zakken van elke variant. Elk monster bestond uit ten minste vijf kernen die genomen waren met een Egner monsternemer (Fig. 23). Er werden monsters genomen tot een diepte van 30 cm. Na het verzamelen werden de bodemmonsters in een vrieskist bewaard en na terugkomst van de excursie diepgevroren. Bodemparameteranalyses zullen later in het laboratorium worden uitgevoerd.

Fig. 23 Bodembemonsteringsschema (boven) en een van de vijf deelmonsters (onder)